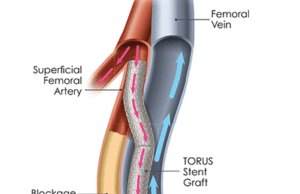
Tag: superficial femoral artery
Real-world registry shows sustained efficacy for Eluvia DES in the SFA
This advertorial is sponsored by Boston Scientific.
During a satellite symposium session dedicated to real cases and trial outcomes which was held at the Cardiovascular...
Pilot study seeks to highlight BEST endovascular strategy for complex lesions...
At this year’s Leipzig Interventional Course (LINC 2023; 6–9 June, Leipzig, Germany), Sabine Steiner (Leipzig, Germany) shared first results from the BEST superficial femoral...
S.M.A.R.T. Radianz vascular stent system approved for transradial use in the...
Cordis recently announced that the US Food and Drug Administration (FDA) has approved the S.M.A.R.T. Radianz vascular stent system, a self-expanding stent purposefully engineered...
Viabahn use in claudicants with long, complex SFA lesions “safe and...
Recently published research indicates that stent grafting with the Viabahn endoprosthesis (W L Gore & Associates) of long and complex superficial femoral artery (SFA)...
Cook Medical receives FDA approval for first 5mm diameter SFA drug-eluting...
Cook Medical have announced that a new 5mm diameter version of Zilver PTX was approved by the US Food and Drug Administration (FDA). It...
IN.PACT Admiral DCB launched in Japan
Medtronic plc has announced the full commercial launch of the IN.PACT Admiral drug-coated balloon (DCB) in Japan. The launch follows the completion of a...
DETOUR I 12-month data show promising durability of bypass system
Results from the DETOUR I trial evaluating the DETOUR System (PQ Bypass) for percutaneous bypass showed promising 12-month durability for patients with extremely long...
LINC 2018 data highlight IN.PACT Admiral as safe and effective treatment...
Medtronic added to its body of clinical evidence supporting the IN.PACT Admiral drug-coated balloon with new presentations that demonstrated durable and consistent clinical outcomes...
Positive results for Boston Scientific Ranger paclitaxel-coated PTA balloon catheter
Positive results from the RANGER SFA trial for Boston Scientific’s Ranger paclitaxel-coated percutaneous transluminal angioplasty (PTA) balloon catheter have been presented at the 2017...
Leaving nothing behind: Is it time to move away from primary...
This educational supplement is only available in countries in Europe.
This educational supplement explores the concept of “leaving nothing behind" in the treatment of the superficial...
Medtronic launches European below-the-knee clinical study of drug-coated balloon
Medtronic has launched the IN.PACT BTK study to evaluate the effectiveness of using a drug-coated balloon in patients with below-the-knee (BTK) peripheral artery disease.
This...
TIGRIS trial finds no stent fractures and positive primary patency in...
Twenty-four month TIGRIS trial follow-up data show that treatment using the Tigris nitinol stent (Gore) for long lesions in the superficial femoral and popliteal...
QT Vascular announces FDA approval to initiate pivotal trial of Chocolate...
The US Food and Drug Administration (FDA) has granted QT Vascular conditional Investigational Device Exemption (IDE) approval, allowing the company to begin enrolling patients...
Gore Viabahn endoprosthesis celebrates 20 years since introduction
Gore is celebrating the 20th anniversary of the introduction of the Viabahn endoprosthesis; its stent graft for the treatment of complex peripheral vascular disease....