Tag: DETOUR
Endologix initiates postmarket study of the Detour system
Endologix has announced the initiation of the Percutaneous transmural arterial bypass (PTAB)1 postmarket study. This study marks the beginning of a comprehensive postmarket study...
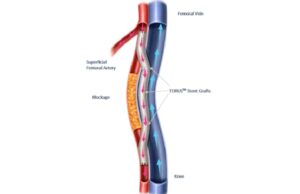
Endologix announces completion of enrolment in TORUS 2 study for PAD...
Endologix has completed enrolment in the TORUS 2 investigational device exemption (IDE) clinical study in the USA, a press release reports.
The TORUS 2...
DETOUR I two-year outcomes: “Excellent” functional improvement in complex PAD cohort
During a late-breaking data session at this year’s Vascular Interventional Advances annual meeting (VIVA 2020; 6–8 November, virtual), Ehrin Armstrong (University of Colorado, Denver,...
First patient treated in DETOUR II IDE trial
PQ Bypass has announced the enrolment of the first patients in the pivotal DETOUR II trial in the USA. The trial is a prospective,...
PQ Bypass receives IDE approval to initiate pivotal DETOUR II clinical...
PQ Bypass has received conditional approval of its investigational device exemption (IDE) from the US Food and Drug Administration (FDA) to initiate the pivotal...
PQ Bypass reports positive results for Detour system in patients with...
A subset analysis of the DETOUR I clinical trial showed promising safety and effectiveness results of PQ Bypass’ Detour system for treating long-segment (>25cm)...
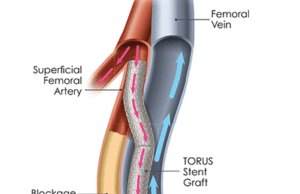
PQ Bypass announces CE mark for Detour percutaneous bypass technologies
PQ Bypass has announced CE mark approval for a trio of proprietary devices: the Torus stent graft system, PQ Snare, and PQ Crossing Device,...