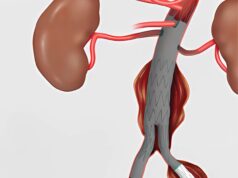
Gore has received FDA approval for the 23mm and 27mm diameter sizes of the contralateral leg component of the Gore Excluder endoprosthesis for treatment of abdominal aortic aneurysms. The new diameter devices provide physicians with the ability to repair aneurysms in a wider range of anatomies eligible for EVAR.
“By adding new diameter options to the Gore Excluder device, patients with large iliac arteries can now be treated with fewer components. This will simplify the EVAR procedure for these patients, widen its applicability, and reduce its costs,” said Michel Makaroun, professor and chair, Division of Vascular Surgery, University of Pittsburgh School of Medicine, Pittsburgh, USA.
Once the physician has positioned the Excluder in the diseased aorta, the Gore C3 Delivery System intuitively enables repositioning of the stent graft. The ability to reposition the device may minimise complications that could occur if the graft needs to be moved after the initial deployment.
“The availability of new diameter Gore Excluder device components expands the use of this device to a wider range of anatomical considerations,” said David Abeyta, Gore Aortic Business Leader.