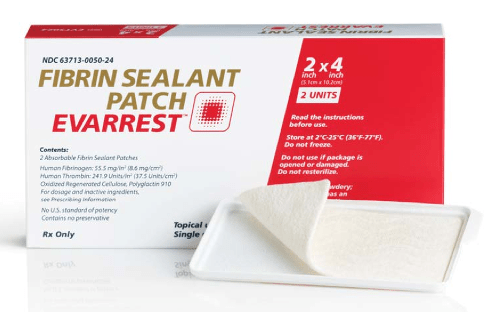
Ethicon has announced new data from a global cardiovascular Phase III clinical trial of the Evarrest fibrin sealant patch. Seventy five per cent of patients treated during aortic reconstruction surgery with Evarrest achieved haemostasis on the first attempt within three minutes and maintained haemostasis throughout the duration of the surgery.
Results from this investigational study were presented at The Houston Aortic Symposium (3–5 March, Houston, USA).
The Evarrest cardiovascular Phase III study concludes a series of pivotal trials designed to demonstrate the fibrin sealant patch’s safety and efficacy in a variety of tissue types. This latest evidence is part of a pending regulatory submission to the US Food and Drug Administration (FDA) to expand the indication for Evarrest in the USA.
Evarrest is currently approved for use with manual compression as an adjunct to haemostasis for control of bleeding during adult liver surgery and soft tissue bleeding during open retroperitoneal, intra-abdominal, pelvic, and non-cardiac thoracic surgery when control of bleeding by standard surgical methods of haemostasis (eg., suture, ligature, cautery) is ineffective or impractical. If approved by the FDA, Evarrest could be used as an adjunct for general haemostasis.
“Controlling bleeding in a highly complex surgical procedure, like aortic reconstruction, can be challenging,” said Sina Moainie, cardiothoracic surgeon at St Vincent Heart Center of Indiana in Indianapolis, USA, and one of the study’s investigators. “Once again, Evarrest demonstrated positive results, even under the most challenging circumstances such as when applied to a prosthetic graft while on cardiopulmonary bypass and hypothermia.”
The objective of this randomised multicentre Phase III clinical study was to evaluate the haemostatic efficacy and safety of Evarrest as an adjunct to conventional haemostasis for anastomotic suture line bleeding during aortic reconstruction surgery. A total of 156 patients from North America, Europe and Asia were randomised to either Evarrest or TachoSil (Baxter). In this study, the treated target bleeding sites were aortic graft anastomotic suture line bleeding identified upon cardiac reperfusion and while on cardiopulmonary bypass. Haemostasis at the target bleeding site was assessed at three, six and ten minutes, and then up to the time of chest closure. Patient age ranged from 24 to 83 years with 117 male patients and 39 females.
In the intent-to-treat primary endpoint analysis, 57 (75%) patients treated with Evarrest achieved haemostasis within three minutes of product application. Haemostasis was maintained through the initiation of chest wall closure. Thirty six (45%) of those treated on the TachoSil arm achieved the primary endpoint. In the secondary endpoint analysis, 77.6% and 84.2% of patients treated with Evarrest achieved haemostasis at six minutes and ten minutes, respectively, and maintained haemostasis through initiation of chest wall closure. In the TachoSil arm, 56.3% and 70% of patients achieved haemostasis at six and ten minutes, respectively, and maintained haemostasis through initiation of chest wall closure.
No major safety concerns were identified during the study.
“This study adds to the increasing body of evidence on Evarrest and its use to control problematic bleeding situations in even the most challenging patients and surgical situations,” said Richard Kocharian, franchise medical director, Ethicon.