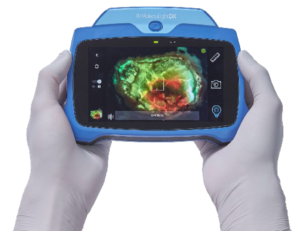
MolecuLight has announced the launch of the MolecuLightDX, a new point-of-care device model targeted at the needs of new expanding wound care market segments in the USA. The DX is an expansion of MolecuLight’s product line and compliments the MolecuLight i:X, a press release reads.
“The i:X and DX are the only commercially-available point-of-care devices to enable real-time detection of elevated bacterial burden in wounds. With the introduction of the MolecuLightDX, we are thrilled to expand our product line and provide added functionality for these wound care market segments,” says Anil Amlani, MolecuLight’s CEO.
Specifically, the MolecuLightDX has the following new features frequently required in these segments:
• Comprehensive electronic medical record (EMR) integration options for multiple EMR environments
• Patient-centric user interface and workflow to allow for easy patient and wound tracking
• An administrator workflow and system configuration capability
• Docking system for easier charging of the device
As with the i:X, the DX has the same accurate, rapid digital wound measurement for documentation of procedures and of wound progression, MolecuLight claims, adding that a stickerless measurement capability which automatically measures wound area without the need for wound stickers is newly available on the DX.
“With the expansion of our product line, we can now offer clinicians in any care setting the unmatched capabilities of the MolecuLight platform, with a feature set and price point that matches their specific needs”, says Amlani. “MolecuLight customers will continue to receive our comprehensive activation and training support on both platforms, including on-site training with patients, e-Learning courses and certification and ongoing in-person and remote support by our Clinical Applications team.”
In addition, all MolecuLight procedures will be able to benefit from the reimbursement pathway available in the USA for the MolecuLight procedure, which is applicable to both the MolecuLight i:X and DX devices. The reimbursement pathway includes two current procedural terminology (CPT) codes for physician work to perform “fluorescence wound imaging for bacterial presence, location, and load” and facility payment for Hospital Outpatient Department (HOPD) and Ambulatory Surgical Center (ASC) settings through an Ambulatory Payment Classification (APC) assignment.
The MolecuLightDX has received US Food and Drug Administration (FDA) clearance for sale in the USA, as well as the CE mark and Health Canada approval for commercial availability in Europe and Canada.
The MolecuLightDX will be displayed in the MolecuLight exhibit booth at the upcoming Symposium on Advanced Wound Care (SAWC) Fall 2021 (29–31 October, Las Vegas, USA).













