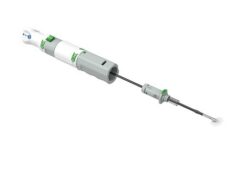
Medeon Biodesign has received approval by the New Zealand Health and Disability Ethics Committees, Ministry of Health to commence the CE clinical trial for the XPro suture-mediated vascular closure device system (XPro system), the company’s large bore closure device, at Auckland City Hospital, New Zealand.
The clinical study will assess the safety and effectiveness of the XPro system to facilitate haemostasis in patients undergoing percutaneous endovascular procedures utilising 8–18F introducer sheaths via the common femoral artery, including endovascular aneurysm repair, thoracic endovascular aneurysm repair, and percutaneous balloon valvuloplasty procedures. The prospective, multicentre, single-arm study will enrol up to 60 patients in New Zealand and Australia. The results will support CE mark regulatory submission.
XPro, the suture-mediated closure device targeting percutaneous large bore procedures, had its safety and effectiveness demonstrated in first-in-man studies in late 2015 at Sanatorio Italiano Hospital in Asuncion, Paraguay. The device achieved immediate or rapid haemostasis at the puncture sites with excellent clinical outcomes, including fast ambulation and discharge from the hospital.
Mark Webster, an interventional cardiologist at Auckland City Hospital, who is also the principal investigator of this clinical study, stated, “Access site complications from large bore vascular sheaths and valve delivery systems remain a concern, and are still a common cause of patient morbidity and, very occasionally, mortality. A dedicated, large bore vascular closure device with reproducible safety and efficacy has the potential to significantly lower the time, effort, and expense of these procedures, as well as improve patient outcomes.”
“Optimal large bore vascular closure can be achieved by three simple steps with the XPro system. Our suture mediated technology with built-in safety mechanisms is designed to assist interventionalists to manage procedures more efficiently while minimise potential complications associated with vessel narrowing and concerns arising from other technologies that leave behind foreign materials in the blood vessel. The commencement of this clinical study will accumulate more clinical experience to demonstrate its safety, effectiveness, and reproducibility of this disruptive technology,” says Yue-Teh Jang, chairman and chief executive officer of Medeon Biodesign.