Lombard Medical and MicroPort Scientific Corporation have finalised the definitive agreements to their strategic partnership first announced on 19 December 2016.
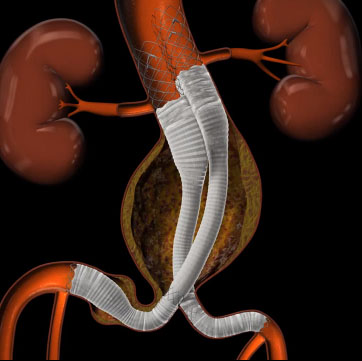
The partnership will allow Lombard to accelerate commercialisation in China and other global markets for its abdominal aortic aneurysms product portfolio: Aorfix, the only stent graft to hold global approvals to treat abdominal aortic aneurysms with aortic neck angles up to 90 degrees, and Altura, an innovative stent graft and ultra-low profile delivery system that offers an easy to use and predictable treatment option for standard abdominal aortic aneurysms anatomy.
Lombard and MicroPort have entered into a component supply manufacturing agreement whereby MicroPort will manufacture, in its facilities in Shanghai, certain components for the Aorfix and Altura product lines. It is anticipated that MicroPort will begin providing components to Lombard in the second half of 2017.
MicroPort has the exclusive marketing rights for the Lombard product portfolio for China and Brazil as well as a technology license to manufacture the products for the China market. MicroPort expects to launch Aorfix in China after gaining China Food & Drug Administration approval which is anticipated in the second half of 2018.
“This strategic partnership begins to establish the foundation for improved gross margins for Lombard by significantly reducing the manufacturing costs of both Aorfix and Altura devices which, in turn, will allow Lombard to be competitive in all of our markets,” Lombard chief executive officer Simon Hubbert commented. “Moreover, given MicroPort’s exceptional market reach, we have laid the groundwork for the Lombard products to enter the fast-growing China endovascular market.”
MicroPort Chairman and chief executive officer Zhaohua Chang stated, “Completing the agreements to the strategic partnership represents an important milestone achieved for both companies so that we can now focus our efforts on the regulatory and operational steps necessary to launch these exciting products in China. We have already been working collaboratively to generate manufacturing synergies and to improve manufacturing efficiency, thereby benefitting the full range of stakeholders in our respective companies. We look forward to bringing the exciting potential of Aorfix and Altura devices to the China endovascular market.”












