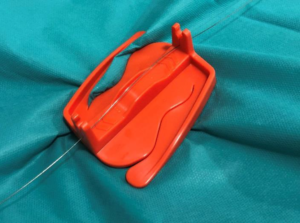
Vascular Barcelona Devices (VB Devices) recently announced that its Crabclamp product received CE mark approval as a Class 1s medical device.
According to a press release, the Crabclamp is a unique device that allows surgeons and their assistants to easily secure guidewires and catheters during endovascular procedures. The device facilitates procedures by avoiding the risk of guidewire loss or untimely movement, while contributing to keeping an orderly surgical field.
The Crabclamp is particularly useful in procedures in which surgeons have limited assistance or those which may be particularly complex, for example, in endovascular aneurysm repair (EVAR) procedures, where two femoral accesses are used. In these cases, it is essential to keep one of the guidewires stable while working with the other access.
Having secured initial distribution agreements with specialised companies in the field, VB Devices will launch the Crabclamp in Europe in January 2021, while continuing to expand its distribution network globally.
“I am excited to see this need-based invention reach the market and hopeful it will facilitate surgical procedures for many of my colleagues and their assistants,” said Enric Roche (Hospital Universitari Sagrat Cor, Barcelona, Spain), co-founder of VB Devices.













