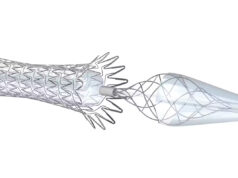
Contego Medical has announced that it has received CE mark for its Vanguard IEP peripheral balloon angioplasty system with integrated embolic protection. The company intends to launch the Vanguard IEP system in Europe to endovascular specialists performing lower limb angioplasty procedures.
The Vanguard IEP system represents the latest innovation of Contego’s integrated embolic protection (IEP) technology, incorporating a peripheral angioplasty balloon and distal embolic filter on the same catheter. The system protects the lower limbs during angioplasty without the need for additional devices or exchanges. The Vanguard IEP system has an over-the-wire design with a sheathless integrated 150-micron pore filter distal to the angioplasty balloon. The Vanguard IEP system is the first filter to feature in vivo adjustability to suit varying vessel sizes and maximise capture efficiency.
“The CE mark for the Vanguard IEP system expands our product portfolio of novel devices designed to provide enhanced safety during peripheral vascular procedures,” said Ravish Sachar, chief executive officer and founder of Contego Medical. “We are eager to positively impact this growing patient population, with an estimated 200 million people affected by peripheral artery disease worldwide.”
“The Vanguard IEP system is an important step toward enhancing safety for patients undergoing peripheral angioplasty for occlusive disease,” said Thomas Zeller, director of the Department of Angiology at Universitaets-Herzzentrum Freiburg in Bad Krozingen, Germany, and principal investigator of the upcoming Vanguard clinical study. “This device will be of particular importance to patients at high risk of distal embolisation, such as those with acute limb ischaemia or chronic total occlusions, as well as those at higher risk should embolisation occur, such as patients with critical limb ischaemia and diabetes mellitus.”