Tag: VIVA 2020
Paclitaxel situation “crystallising” thanks to new data and teamwork, VIVA audience...
This year's Vascular Interventional Advances annual meeting (VIVA 2020; 6–8 November, online) opened with a session on controversial hot topics and advanced multidisciplinary approaches...
ILLUMENATE four-year pooled analysis has “brought increased clarity” to the paclitaxel...
Sean Lyden (Cleveland, USA), current VIVA Board president, speaks to Vascular News about his two years at the helm, which began with the “controversy on...
AngioDynamics announces presentation of positive safety, efficacy results from RAPID outcomes...
AngioDynamics has announced the safety and efficacy results from the RAPID (Registry of AngioVac system procedures in detail) database. Results were shared by principal...
Six-month data from Surmodics Avess AV fistula DCB first-in-human study presented...
Surmodics recently announced that six-month data from the AVESS first-in-human (FIH) study of the company’s Avess arteriovenous fistula (AVF) drug-coated balloon (DCB) was shared...
No safety concerns and favourable patency at two years with Absorb...
The Absorb bioresorbable vascular scaffold (BVS; Abbott Vascular) can be used for the treatment of chronic limb-threatening ischaemia (CLTI) patients in infrapopliteal arteries with...
DETOUR I two-year outcomes: “Excellent” functional improvement in complex PAD cohort
During a late-breaking data session at this year’s Vascular Interventional Advances annual meeting (VIVA 2020; 6–8 November, virtual), Ehrin Armstrong (University of Colorado, Denver,...
VOYAGER PAD supports “pharmaco-invasive” approach to optimise outcomes
Recent data from VOYAGER PAD showed no association between drug-coated device (DCD) use and mortality. Given the absence of a safety signal, the current...
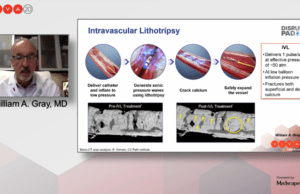
DISRUPT PAD III: Intravascular lithotripsy superior to PTA in acute procedural...
The DISRUPT PAD III randomised controlled trial (RCT) provides the largest level one evidence for the treatment of heavily calcified femoropopliteal arteries, noted William...
Four-year results from patient-level meta-analysis confirm safety profile of Philips Stellarex...
Royal Philips today announced the results of a patient-level meta-analysis that confirms the safety profile of its Stellarex drug-coated balloon (DCB) at four years....
REALITY study “sets new benchmark” for assessment of claudicants with severe...
Krishna Rocha-Singh (Springfield, USA) speaks to Vascular News about his presentation at VIVA20 (Vascular Interventional Advances; 6–7 November 2020; virtual) on the REALITY study,...
PROMISE I: LimFlow system enables vast majority of patients to avoid...
LimFlow SA has announced the presentation of one-year data from the full patient cohort in its PROMISE I study of the LimFlow percutaneous deep...
Similar outcomes for ultrasound-assisted and standard thrombolysis in SUNSET sPE
Efthymios Avgerinos (University of Pittsburgh, Pittsburgh, USA) presented results from the SUNSET sPE trial during a late-breaking data session at this year’s Vascular Interventional...
Safety and efficacy of IN.PACT Admiral sustained out to five years
In a late-breaking data session at this year’s Vascular Interventional Advances conference (VIVA 2020; 6–8 November, virtual) Thomas Zeller (University Heart Center Freiburg, Bad...
One-year VIVO results support safety and effectiveness of Zilver Vena venous...
Results of the VIVO clinical study support the safety and effectiveness of Cook Medical's recently FDA-cleared Zilver Vena venous stent for the treatment of...
Directional atherectomy prior to IN.PACT DCB effective out to one year,...
A vessel preparation treatment strategy of directional atherectomy prior to drug-coated balloon angioplasty with Medtronic’s IN.PACT Admiral drug-coated balloon (DCB) in long, calcified femoropoliteal...