Tag: Vanguard IEP
Contego Medical receives 510(k) clearance for Vanguard IEP system
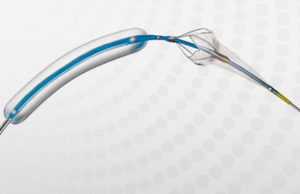
Contego Medical has announced that the US Food and Drug Administration (FDA) has granted 510(k) clearance for its Vanguard IEP Peripheral Balloon Angioplasty System...
Contego Medical announces ENTRAP Study initiation for Vanguard IEP peripheral balloon...
Contego Medical has announced the initiation of the ENTRAP Study evaluating usage of its Vanguard IEP peripheral balloon angioplasty system with integrated embolic protection...