Tag: catheter-directed thrombolysis
Surmodics receives US FDA 510(k) clearance for Pounce XL thrombectomy system
Surmodics has announced that it has received US Food and Drug Administration (FDA) 510(k) clearance for its Pounce XL thrombectomy system.
The Pounce XL thrombectomy...
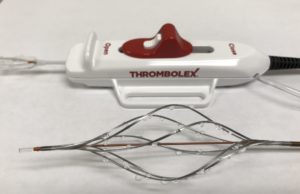
Thrombolex announces new insights from the RESCUE trial with the Bashir...
Thrombolex has announced never-before-reported major reductions in obstruction in all of the segmental pulmonary arteries (PA), based on independent core lab data analysis of...
New trial planned to rigorously test catheter-directed thrombolysis for submassive PE...
Clinically relevant outcomes are needed to measure catheter-directed therapy (CDT) against anticoagulation alone to treat submassive pulmonary embolism (PE) patients. Further, CDT’s ability to...