Tag: Baylis Medical Technologies
Baylis Medical Technologies launches PowerWire 14 RF guidewire
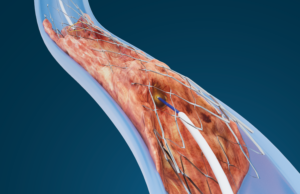
Baylis Medical Technologies today announced US Food and Drug Administration (FDA) 510(k) clearance and commercial launch of the PowerWire 14 radiofrequency (RF) guidewire, designed...
Baylis Medical Technologies introduces PowerWire Pulse radiofrequency guidewire
Baylis Medical Technologies has today announced the commercial launch of the PowerWire Pulse radiofrequency (RF) guidewire in the USA.
A press release notes that the...
Baylis Medical Technologies announces first commercial use of PowerWire Pro for...
Baylis Medical Technologies today announced the completion of its first clinical use of the PowerWire Pro radiofrequency (RF) guidewire to safely cross a chronically...
Baylis Medical Technologies announces launch of PowerWire Pro RF guidewire
Baylis Medical Technologies today announced the 510(k) clearance and launch of the PowerWire Pro radiofrequency (RF) guidewire in the USA, facilitating venous stent recanalisation...