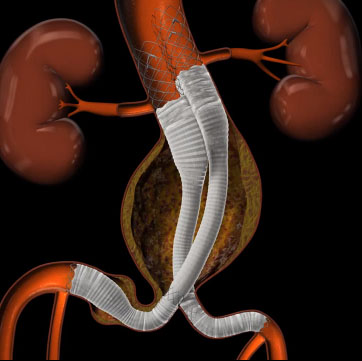
Lombard Medical is to conduct a 1,000 patient global registry to evaluate its new Altura Endograft System. The ALTITUDE registry (AlturaImpact on the Treatment of Abdominal Aortic Aneurysms Using a Novel D-stent EVAR Design) will be conducted outside the USA and will evaluate the use of the Altura endograft stent system in real world patients across a range of clinical centres with a planned enrolment of 1,000 patients starting in late 2016.
The primary investigator will be Paul Hayes, of the Department of Surgery, University of Cambridge and Addenbrookes Hospital, Cambridge, UK.
“With one of the largest AAA patient populations in the UK, we have been privileged to study many new endovascular devices for aortic aneurysm repair,” says Hayes.
“The Altura device represents one of the most exciting new endograft designs in recent memory. With a repositionable aortic section, the elimination of the contralateral cannulation step and retrograde delivery of the limb sections, this design should facilitate a much simpler, safer and consistent deployment. This simplicity should also help with surgical planning and possibly allow patients to be discharged sooner after surgery. The 14F ultra-low profile delivery catheter can also accommodate smaller access vessels that may be compromised due to disease. I look forward to leading this important new clinical registry and establishing a robust set of clinical data points.”













