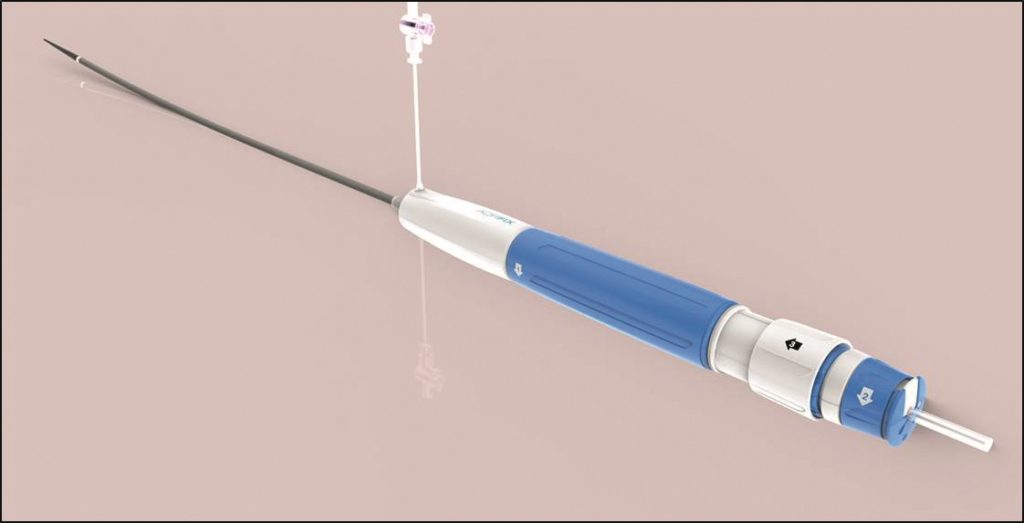
Lombard Medical has announced approval from the Japanese Ministry of Health, Labour and Welfare for its IntelliFlex low-profile (LP) delivery system for the Aorfix endovascular stent graft. Commercial launch will follow reimbursement approval, which is anticipated in February 2017.
Aorfix with IntelliFlex is exclusively distributed by Medico’s Hirata, a Japan’s supplier of vascular products with expertise in building significant market share for abdominal aortic aneurysm stent grafts.
This new delivery system is used in conjunction with the Aorfix endovascular stent graft, the only abdominal aortic aneurysm stent graft with global regulatory approval to treat aortic neck angulation up to 90 degrees. The IntelliFlex LP delivery system features a low-profile, compact and ergonomic design that provides intuitive control of Aorfix during deployment, even in challenging anatomies. The system features an integrated exchange sheath allowing ancillary devices to be introduced during the endovascular procedure, avoiding the need for sheath exchanges thus potentially reducing blood loss, vessel trauma, procedure cost and time.
Masataka Hirata, president of Medico’s Hirata said, “We have enjoyed great success already with Aorfix in Japan and built our market share to approximately 8% over the last two years. Now with the approval of IntelliFlex, we believe we will significantly increase the usage of this unique technology for the benefit of physician’s ease of use, procedural outcomes and patients across the broadest range of abdominal aortic aneurysm anatomies.”
“Japan is the world’s second largest standalone EVAR market and is both strategically and financially important for us,” said Lombard Medical chief executive officer Simon Hubbert. “With our partner, Medico’s Hirata, we look forward to providing Japanese physicians with the Aorfix stent graft, which has outstanding clinical results, now delivered with the unique advantages of the IntelliFlex LP delivery system.”













