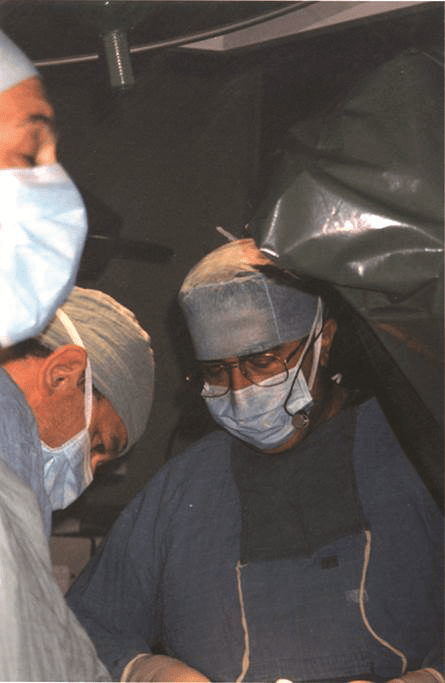
On 7 September 1990, a 70-year-old man with severe chronic obstructive pulmonary disease and severe back pain caused by a 6cm abdominal aortic aneurysm underwent the first successful endovascular aneurysm repair (EVAR) in the Western world. The case by Juan C Parodi and Julio Palmaz became the first widely known endovascular repair of the aorta and was described the following year in the Annals of Vascular Surgery. On the 25th anniversary of that landmark case, we dedicate these pages to some of the EVAR pioneers.
Parodi, honorary professor of Surgery, University of Buenos Aires, Buenos Aires, Argentina, and adjunct surgical research professor, University of Michigan, USA, was honoured in June with the John Homans Distinguished Lecture at the Society for Vascular Surgery (SVS) Vascular Annual Meeting (18–20 June, Chicago, USA). Peter Lawrence, then SVS president, said: “The history of endovascular repair has at its root, Dr Parodi’s inspiration, persistence and innovation, which began when he was a vascular surgery resident at the Cleveland Clinic in the mid-1970s. He returned to Argentina and pursued his new method of treating aneurysms. We are honoured to be celebrating the 25th anniversary of his first endovascular repair of an abdominal aortic aneurysm.”
Parodi’s journey towards an endoluminal repair for aortic aneurysms began when he was 34 years old. After he and his team had two consecutive bad outcomes following abdominal aortic aneurysm repair on the same day, Parodi was inspired to develop a new approach. Being trained on the Seldinger technique he suggested to his boss Alfred Humphrey: “Al, I think we can do the Seldinger technique with a bigger catheter, introduce a compressed graft and a metal component to replace the suture [and achieve,] all this transfemorally.” That was the beginning of Parodi’s quest for a less invasive, less morbid, less lethal and equally effective procedure.
The conception of the endoluminal technique started in 1976. It was aimed at excluding an aneurysm using a fabric graft and metal components to affix and seal these elements in position using an endovascular retrograde access. At that time, the term “metal cage with barbs” was used rather than “stent”. The engineering of the first prototypes using elastic wires of stainless steel and the experimental work took place at the Cleveland Clinic, in Cleveland, USA.
In 1979, after seeing a patient become paraplegic after an intervention for a dissection, Parodi presented the first protocol to treat type B aortic dissections endoluminally. The idea was to cover only the entry site with the cylindrical covered metal he had developed. However, the project was rejected and received no support.
A few years later, Parodi met Argentinian radiologist Julio Palmaz, the inventor of the Palmaz stent. “I thought his stent could be better than mine and I started using it. I redesigned the stent so it would go up to 30mm and started using it in animals in 1987.”
From 1976 to 1990, Parodi did not receive any funding or support for his project. “We were using our own money for this development. We were chasing dogs on the streets of Buenos Aires, carrying a syringe with Penthotal. We were taking these dogs to the hospital, with permission from the director to replace their aorta with a graft in the shape of an aneurysm, and after a few months we were ready to put the endograft in. In total, we operated on 53 dogs, followed them for six months at the farm of one of my friends, and then did an anatomic-pathological study of the devices. The good results encouraged us to do the first human case. Finally, my institution gave me permission to do 50 cases in humans on the condition that they had to have been rejected for intervention previously by at least two centres,” he says.
In 1990, Parodi received a phone call from the president of Argentina. The president told Parodi: “My secret service has told me you are doing something new for aneurysms. My cousin has an aneurysm, back pain and severe chronic obstructive pulmonary disease, so please use it in him.” The patient and his family went to see the surgeon. Parodi remembers: “I explained the procedure, showed angiograms of before and after, the procedure and they said, ‘You have a lot of experience’. I said, ‘Wait, those are dogs. You are going to be the first human!’.”
Parodi went ahead with the operation and the president’s cousin became his first successful EVAR case. “We completed the procedure under local anaesthesia and he did very well,” said Parodi. Three months after the first intervention, the patient developed a distal endoleak since Parodi had not placed a distal stent. He explained that several patients with aorto-aortic devices required a second intervention due to dilatation or endoleak of the distal end. Those patients were treated with aorta-uni-iliac endografts, occlusion of the contralateral common iliac and a femoro-femoral bypass, a method that provided “excellent results”. Parodi’s first patient lived for nine years after the procedure when he developed pancreatic cancer.
The first device was simple, according to Parodi. He comments: “It was a graft I designed with expandable ends, the extra-large Palmaz stent, a Teflon sheath with a valve, a wire, and the valvuplasty balloon, which I took from the cardiologists. For the first cases, I invited Palmaz, who assisted me. We did the first endoluminal case, and the following case that day was an open repair case. We then stopped, had dinner and after dinner we went back to see our patients. The patient who had had the endoluminal treatment under local anaesthetics was having dinner and the other one was still intubated. Julio told me: ‘If we can prove that we can repeat this procedure and it is durable, we are going to start a revolution’. At that time we created a hybrid, new speciality. At that point it became clear that we had to learn about catheters, wires and stents.”

After a few months, Parodi started to see complications in the first patients treated with EVAR. He explains: “The failures were related to the aorto-aortic system. Because the distal neck is sometimes non-existent, or very short, we were having leaks in the distal part. We then decided to create a bifurcated graft. We did it, placed the stents and the angiogram looked beautiful, but unfortunately the basic success was -23 and the patient had a massive microembolization. The excessive instrumentation caused the first fatal complication. I was very disappointed and stopped the project. In a few months, we restarted, with high-risk patients, doing aorto-uni-iliac grafts in 1991, occluding the contralateral limb with the detachable balloon initially and with covered stents afterwards. The device was very primitive, but it worked.”
Parodi said: “I was foreseeing a day when the patient would walk into the operating room, we would carry out the case under local anaesthesia, percutaneously, place a bandage and the patient would walk home the same day. As a matter of fact, now we do ambulatory surgery in one-third of our patients.”
Parodi and his team had to come up with solutions for the different challenges the endovascular approach imposed. When there was a lack of distal aortic neck, an aorto-uni-iliac endograft was used. When the problem was a narrow external iliac artery, a temporary conduit using a 10mm Dacron graft was created. In the case of an iliac aneurysm with a jeopardised internal iliac artery, embolization of the internal iliac artery was performed or, in most cases, an anastomosis between the external iliac artery and the hypogastric artery was performed.
The first five cases, from Argentina, were published by Parodi, Palmaz and Barone in Annals of Vascular Surgery in 1991. In Western Europe, the first cases were performed in October 1992 by Parodi, Claude Mialhe, Claude Amicabile and Claudio Schonholz in Nancy, France; in the USA, the first cases were performed in 1992 by Parodi, Frank Veith, Michael Marin, Jacob Cynamon and Claudio Schonholz in New York.
Across the globe, several vascular specialists were seeking a less invasive treatment for the problems of the aorta. “There were many people thinking the same way we did—Choudhury, Kronemberg, Balko, Lazarus, Minch and Volodos. We were very persistent,” notes Parodi. “We opened our operating room up to anybody who wanted to learn. I travelled the world training colleagues, presented the idea openly in all meetings without any monetary compensation; it was a crusade to change the specialty in order to survive and prevail,” says Parodi. “Since then, the field has come a long way and we can now use stent grafts for aneurysms, dissections, vascular trauma and occlusive disease. I have to pay tribute to Roy Greenberg and Timothy Chuter as they went beyond and developed something to cover the whole aorta. Chuter developed fenestrated and branched devices, and procedures using these types of devices are now routinely performed. Roy Greenberg developed the helicoidal hypogastric branch and Cook the straight one.”
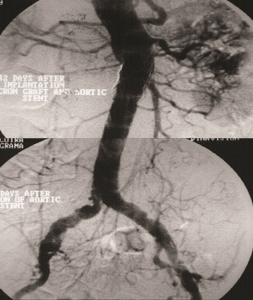
Parodi also noted that since the introduction of EVAR, the annual number of deaths from intact and ruptured aneurysms has significantly decreased. He quoted Giles et al (JVS 2009), saying that “this coincided with an increase in intact aneurysm repair and a decrease in ruptured aneurysm diagnosis and repair volume.” A great indication that has emerged more recently is the treatment of ruptured aneurysms, with encouraging results, Parodi said. Ben Starnes, for example, has a 16% mortality rate in all-comers, including para-renal aneurysms.
To demonstrate that EVAR is durable, Parodi showed the 20-year follow-up of a patient treated with an aorto-aortic stent graft, the “initial primitive device”, in Europe in 1994. “A CT scan was sent to me last year. The balloon-expandable device did not migrate and did not produce any leaks.”
Data from the US Census Bureau show that there were 101,978 patients in the National Inpatient Sample affected by abdominal aortic aneurysms over the 11-year span of this study (JVS 2014; 59:1512-1517). The average age was 73 years, 21% were women and 90% were white. Overall in-hospital mortality was 7%, with a median length of stay of five days and median hospital charges of US$58,300. EVAR was associated with a lower in-hospital mortality rate (4% vs. 1% for unruptured and 41% vs. 27% for ruptured aneurysms, p<0.001), shorter median length of stay (seven vs. two; nine vs. six, p<0.001) but a 27–36% increase in hospital charges. In hospital mortality rates for both ruptured and unruptured cases have fallen by more than 50% during this time period (2000–2010).
“Most of what we do today in vascular surgery is different from what we learnt in our training. Minimally invasive procedures are now part of the armamentarium of vascular surgery. Vascular surgeons embraced minimally invasive procedures very early and had to make the effort to learn these new techniques. Had they remained exclusively committed to open surgery, vascular surgery would not exist today. Cardiac surgeons did that and as a consequence, they are doing fewer minimally invasive procedures than vascular surgeons,” Parodi told delegates at the SVS annual meeting. “EVAR was the wake-up call for most vascular surgeons. Iliac artery and superficial femoral artery stenting are commonplace now. We also do carotid, subclavian and visceral stenting. The arguments against EVAR were instrumental in prompting new and improved devices. Fenestrations and branched endografts further extended our practice. TEVAR has allowed us to cover aneurysm, dissections and trauma of the aorta.”
Parodi concluded: “We still have problems—large neck aneurysms, cases outside the instruction for use, type II endoleaks and ruptures—and our journey still continues. I believe we are solving these issues progressively and I am very optimistic we are doing better every day.”
“Each case presented new challenges and new solutions had to be created”
By Claudio Schonholz
When I met Juan Parodi in 1989 he was very close to achieving his dream of treating patients with abdominal aortic aneurysm using a minimally invasive, endovascular technique to exclude the aneurysm from the circulation and prevent rupture. He came up with this idea back in 1976 while he was a resident at the Cleveland Clinic and observed a high morbidity and mortality rate in patients with abdominal aortic aneurysms treated by open surgical operation. In that first encounter together, after we reviewed the images of a biliary procedure done on one of his patients, Juan shared with me some of the details of his project. He got my full attention since he was proposing an endovascular alternative to an otherwise very invasive treatment.
At that time and after my fellowship in Vascular and Interventional Radiology at the Centre Cardiologique du Nord in Paris, France, I was dedicated to developing a centre for cardio, peripheral and neurointerventional procedures with a group of radiologists, neurosurgeons and interventional cardiologists. Inspired by the initial work done by Jack Theron, we were doing carotid angioplasty under cerebral protection with distal balloon occlusion in patients with high-grade carotid disease prior to coronary artery bypass. I shared images of these procedures with Juan and I got his attention but in an unexpected way since he was very concerned about the risk of distal embolization and stroke. He suggested that I stop this “very dangerous procedure” and invited me to join his project.
The results of the animal studies done both in San Antonio, USA, and Buenos Aires, Argentina, were very encouraging, and in September 1990 Juan Parodi was asked to treat a high-risk patient with an infrarenal abdominal aortic aneurysm that had the perfect anatomy (proximal and distal aortic necks) for the aorto-aortic stent graft device that he and Hector Barone had put together for the occasion. Juan invited Julio Palmaz to participate in the case which took place at the Instituto Cardiovascular de Buenos Aires. That case was a turning point in the history of medicine, changing the way most patients with aneurysmal disease are treated around the world today.
A new technique was born. With each new case challenges surfaced during the procedure and solutions were created to overcome difficult anatomy, the lack of experience with this new technique, and the primitive technology. Everything was new: which catheter [to use], which wire [to use], how to navigate a large device in tortuous anatomy, how to measure the length of the aorta in order to tailor the right device for that patient. It was an exciting time and a privilege to share that seminal work with Juan, who always provided solutions and ways to improve the technical challenges of a completely different technique. We were motivated by the excellent clinical results of this high-risk group of patients which benefited from the initial steps of the endovascular revolution.
Scientific presentations in meetings and the landmark publication in the Annals of Vascular Surgery contributed to the dissemination of the technique, and in October of 1992 Juan Parodi and the team were invited by two interventional cardiologists involved in endovascular peripheral interventions, Michel Henry and Max Amor, to perform a live EVAR case during their meeting in Nancy, France. We were introduced to Claude Mialhe and Claude Amicabile and did the first European EVAR together. Mialhe was already involved in the endovascular treatment of aortoiliac disease and was familiar with balloon angioplasty and stent placement. He showed interest in learning every detail of the technique and very soon after that procedure he came up with the idea of building a modular bifurcated stent graft for EVAR which was named Stentor. This concept was the basis of most of the devices that we use today.
In November 1992 the team travelled to New York to perform the first US EVAR following an invitation from Frank Veith and Michael Marin. Their patient was very challenging (obese and oxygen dependent) and we were given a very primitive imaging technology for the procedure. Before we started the procedure the anaesthesiologist informed us that the patient was too sick for general anaesthesia and even for epidural anaesthesia and that we could only proceed with surgery under local anaesthesia. Once again Parodi showed that he had the knowledge, determination and courage required to move forward in order to accomplish his goal. The procedure went well and the patient was sent home without complications and lived for several years with an excluded aneurysm.
During the years that followed, significant contributions were made by many other physicians from different specialties including vascular surgery, interventional radiology, cardiothoracic surgery and interventional cardiology, culminating in the high level of technology that today allows us to treat close to 80% of patients with thoracic and abdominal aortic aneurysm using the endovascular technique. Early on, most of the centres had vascular surgeons with limited or no experience in endovascular techniques who teamed up with interventional radiologists or cardiologists. These multidisciplinary collaborations have been very productive but unfortunately have not continued among many physician groups who have engaged in negative turf battles.
In my case, I continue to enjoy long discussions on the phone with Parodi on how to improve existing technology or create something new, and the group I work with at the Medical University of South Carolina experiences continued success because of our strong multispecialty collaboration. In 1996 Parodi encouraged me to restart the carotid angioplasty programme, and he came up with the idea of flow reversal to protect the brain during carotid artery stenting: one more brilliant idea to allow safe treatment for high-risk patients, in this case with carotid disease. He continues to fine tune his own discoveries to the benefit of patients everywhere and those of us who treat them.