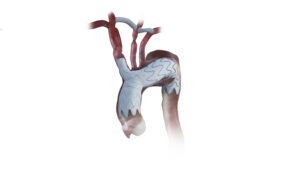
Endospan today announced that the US Food and Drug Administration (FDA) has approved the Nexus aortic arch stent graft.
FDA approval was supported by the one-year results of the TRIOMPHE investigational device exemption (IDE) study, which showed that the Nexus system could effectively treat aortic arch disease, including chronic aortic dissections, in patients at high risk for open surgical repair. The approval enables a US commercial launch of the Nexus system.
Nexus is a bimodular system designed to mimic the ascending and arch anatomy. It has a 20Fr delivery system with a pre-shaped catheter to allow one pass into the arch to minimise arch manipulation and an integrated branch designed for haemodynamic efficiency, Endospan details in a press release.
TRIOMPHE is a prospective, multicentre study designed to assess the safety and effectiveness of the Nexus system in high-risk surgical patients. The one-year results for the high-risk surgical patients with chronic dissections demonstrated safe and effective treatment in the ascending aorta.
“The anatomical design of the Nexus system addresses many of the complexities that occur when treating the ascending and aortic arch,” said Brad Leshnower (Emory University, Atlanta, USA), national cardiac surgery co-principal investigator. “I am pleased to now be able to offer this minimally invasive solution to patients with life-threatening diseases involving the ascending aorta and arch who are at high risk for conventional open repair.”

Ross Milner (UChicago Medicine, Chicago, USA), national vascular surgery principal investigator, added: “On behalf of my co-principal national investigator Dr Leshnower and me, I would like to thank the entire TRIOMPHE investigational body for their commitment, time and effort to make this technology available and expand our treatment options for delivering aortic care to the community.”













