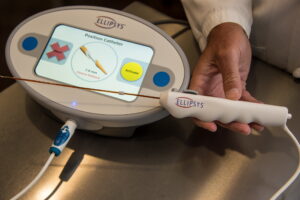
Ellipsys Medical today announced that the Ellipsys vascular access system will once again be commercially available, with initial availability anticipated in mid-2026. The announcement marks the planned reintroduction of the minimally invasive technology to clinicians and patients following a period of limited market availability.
The Ellipsys system, previously available in the USA and internationally, provides a percutaneous approach to creating an arteriovenous (AV) fistula for patients requiring haemodialysis access. Its return “restores access to a technology that many clinicians have continued to seek as interest in less invasive approaches to creating vascular access grows,” a press release reads.
“We are excited to restore access to Ellipsys for clinicians who have continued to advocate for minimally invasive fistula creation,” said Brian Driscoll, CEO of Ellipsys Medical. “The system has a strong clinical foundation, and its return reflects both the demand we’ve heard from the field and our commitment to expanding vascular access options.”
The Ellipsys system is an ultrasound-guided, single-catheter device that enables physicians to create an AV fistula without open surgery or a permanent implant. The procedure can be performed in a hospital, ambulatory surgery centre, or office-based lab, typically using local anaesthesia.
A company press release notes that, since receiving US Food and Drug Administration (FDA) clearance in 2018, the system has been used in thousands of procedures worldwide and has been supported by peer-reviewed publications, multicentre studies, and real-world clinical experience demonstrating high maturation rates and durable outcomes.
“For patients living with kidney failure, every procedure has a meaningful impact on their daily lives,” said Terry Litchfield, president, Access Solutions. “By offering a minimally invasive option that avoids open surgery and supports reliable dialysis access, Ellipsys can help reduce the burden of care and improve the patient experience.”
Ellipsys Medical shares that it plans a phased reintroduction of the system, including the restoration of physician training programmes, clinical education initiatives, and customer support infrastructure across the USA and select international markets.













