
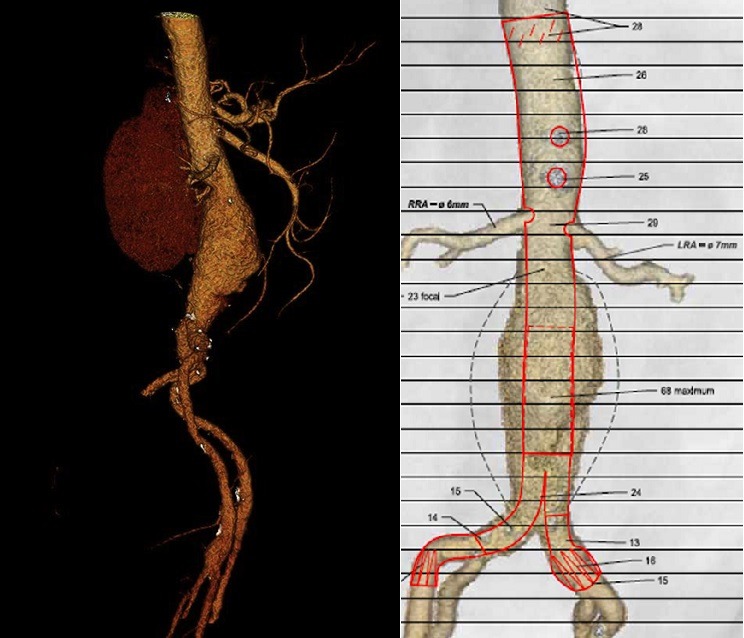
Over the past two decades, endovascular repair of abdominal aortic aneurysms (EVAR) has gained popularity. However, specified anatomic characteristics are required to achieve durable outcomes, and up to 40% of patients are unsuitable candidates for traditional infra-renal EVAR, mainly because of a hostile proximal neck anatomy. To overcome these limitations, there have been continuous improvements in endovascular devices, ancillary equipment, and techniques used to expand anatomic eligibility to a wider number of patients. Stéphan Haulon writes that in aneurysms involving or adjacent to the visceral segment of the aorta, endovascular repair using a fenestrated stent-graft (FEVAR) with three or four fenestrations currently represents the most reliable endovascular option (figure 1).
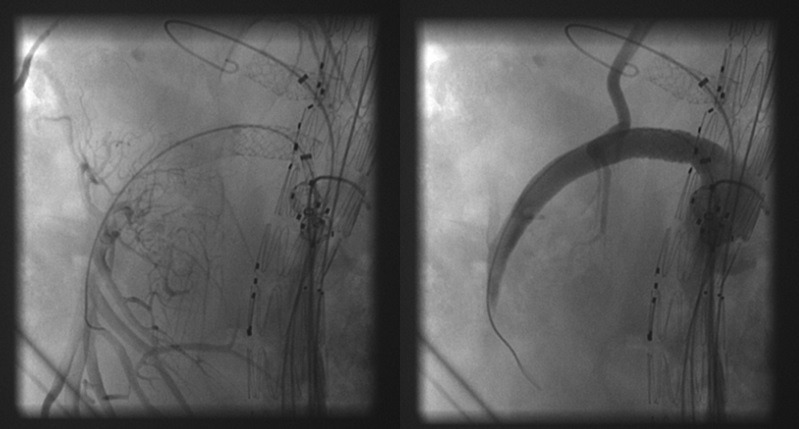
We started using the first generation BeGraft peripheral stent (Bentley InnoMed) in June 2015 in our centre because it was the only covered stent commercially available in large diameters and short lengths (9mm and 10mm diameters, 27mm length, 7F compatible), and thus is perfect for bridging the superior mesenteric artery (SMA) and the celiac trunk (figure 2). In October 2015, Bentley released a second generation covered stent which had received various modifications, including doubling the thickness of the ePTFE-graft material, and increasing the strength of the stent connectors. Its off-label applicability as a bridging stent in aneurysm repair was thus improved.
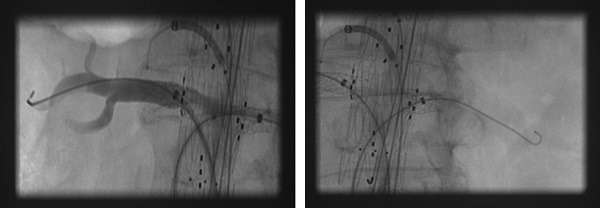
At the same time we started using the preloaded system almost exclusively when performing FEVAR. This system includes two sheaths positioned in the ipsilateral delivery system to create a stable platform to cannulate branches, and provides access to branches from the ipsilateral side (figure 3), limiting contralateral access to the SMA and the celiac trunk. The lack of space inside the introduction system only allows room for two 6F sheaths. The Bentley bridging stents are the only commercially available bridging stents that are 6F-compatible for sizes >6mm in diameter and >22mm in length. When such diameters and lengths are required, the Bentley stent limits the risk of stent dislodgement and type III endoleaks because the appropriately sized stent is placed in the first instance.
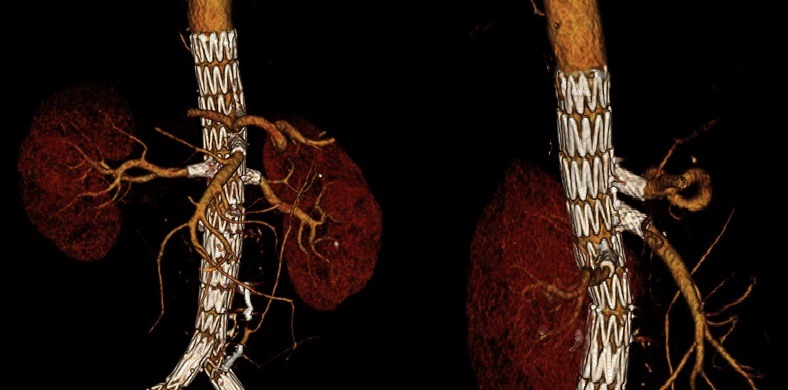
By end of July 2016 we had implanted 92 second generation Bentley bridging stents in 37 patients undergoing FEVAR. To date we have depicted one early occlusion of a target vessel, which resulted from a dissection of the renal trunk distal to the bridging stent. This later patient underwent the only early secondary procedure. To date, no endoleaks related to the bridging stents were depicted during follow-up.
When used as a bridging stent during FEVAR, our initial results show that the Bentley bridging stent is a safe and effective device (figure 4). We restrict its use to fenestrated cases where the fenestrations abut the origin of the target vessels. We have no experience and thus we do not recommend its use in combination with branches.
Stéphan Haulon is at the Hôpital Cardiologique, CHRU de Lille, France. He is a consultant for Bentley InnoMed and Cook Medical













