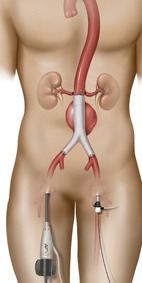
The Australian Therapeutic Goods Administration (TGA) has approved Endologix’ AFX2 bifurcated endograft system for inclusion on the Australian Register of Therapeutic Goods. The TGA has approved the use of AFX2 for the treatment of abdominal aortic aneurysms.
Vikram Puttaswamy, vascular surgeon at Sydney Vascular Surgery in Sydney, Australia, comments, “We are pleased with the approval of AFX2 in Australia and look forward to providing this new technology to our patients. AFX2 and its unique ability to preserve the aortic bifurcation represents an important addition to our endovascular abdominal aortic aneurysm treatment options.”
AFX2 is designed to reduce procedure steps for the delivery and deployment of the bifurcated endograft. The new device is also intended to facilitate percutaneous endovascular aneurysm repair by providing the lowest profile contralateral access through a 7F introducer. These improvements bring together Endologix’s ActiveSeal technology and DuraPly ePTFE graft material into an integrated new endovascular aneurysm repair system.













