This advertorial is sponsored by Gore.

Even though it is celebrating its aortic portfolio reaching its 25-year milestone, Gore believes that this is only the beginning when it comes to being an aortic ally. The GORE® EXCLUDER® AAA Endoprosthesis and the GORE® TAG® Device family are commemorating 25 years of commercial availability and the procedural innovations they have inspired along the way.
Twenty-five years of patient impact and durability is worth celebrating in the life of any medical device. And, when a device reaches this milestone, it is celebrating more than just 25 years in the treatment landscape.
The EXCLUDER® device and the TAG® Conformable Device have been used to treat more than 675,000 patients worldwide and have become the most studied thoracic aortic aneurysm repair (TEVAR) and endovascular aortic aneurysm repair (EVAR) devices according to company-sponsored trials and registries shown on ClinicalTrials.gov of all currently available stent grafts. Contributing to the advancement of aortic treatment with over 1,500 peer-reviewed publications, this is a testament to the device’s legacy—making a difference for physicians and their patients.
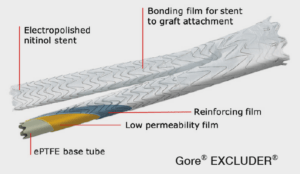
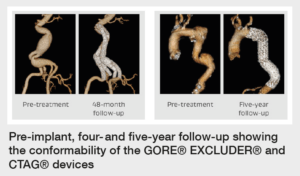 The durability of this device can be attributed to its material quality and design. Both devices feature a nitinol stent, sutureless stent-to-graft attachment, and proprietary expanded polytetrafluoroethylene (ePTFE) film layers.
The durability of this device can be attributed to its material quality and design. Both devices feature a nitinol stent, sutureless stent-to-graft attachment, and proprietary expanded polytetrafluoroethylene (ePTFE) film layers.
“The EXCLUDER® Device has been on the market helping patients for well over two decades—a truly remarkable accomplishment and the longest stretch in the industry,” says Michel Makaroun, chief of the Division of Vascular Surgery at the University of Pittsburgh Medical Center (Pittsburgh, United States) who has also been an investigator in all EXCLUDER® Device clinical studies to date.
“The close collaboration between medical community and manufacturer has allowed for numerous innovations and improvements along the way, providing more accurate deployment and better outcomes.”
 “I have much admired the ongoing family interest in the business and how it has continued in the way that Gore intended it,” says Roger Greenhalgh (London, United Kingdom), the chairman of the Charing Cross (CX) Symposium, of Gore’s history of innovation and research. “The company is known for supporting high-quality medical education.”
“I have much admired the ongoing family interest in the business and how it has continued in the way that Gore intended it,” says Roger Greenhalgh (London, United Kingdom), the chairman of the Charing Cross (CX) Symposium, of Gore’s history of innovation and research. “The company is known for supporting high-quality medical education.”
Over the last two decades, in collaboration with passionate healthcare providers, Gore has continued to develop and study additional solutions within its aortic portfolio, receiving approvals for larger trunk and contralateral limb diameters, and iliac branch devices. As well, the next generations of both devices are conformable and feature the GORE® ACTIVE CONTROL System, allowing physicians more treatment options to consider for their patient’s anatomy.












