This article is an advertorial by Medtronic.
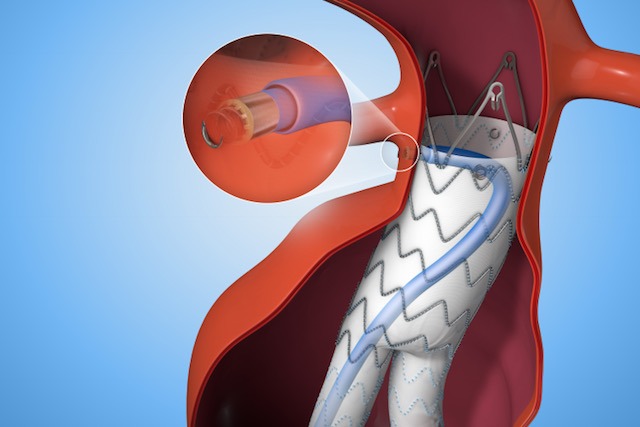
In collaboration with leading clinicians, researchers and scientists worldwide, Medtronic Inc. offers the broadest range of innovative medical technology for the interventional and surgical treatment of cardiovascular disease. One of these innovations, the Endurant™ II/IIs Stent Graft System, is built on Medtronic’s 18 plus years of aortic stent graft experience and is proven in more than 300,000 implants worldwide[1].
Medtronic received approval from the U.S. Food and Drug Administration (FDA) in October 2017 and CE (Conformité Européenne) Mark in December 2017 for the Endurant™ II/IIs stent graft system to treat abdominal aortic aneurysm (AAA) patients with neck lengths less than 10mm and down to 4mm when used in combination with the Heli-FX™ EndoAnchor™ system. The expanded indication enables the Endurant™ II/IIs stent graft to be used in conjunction with the Heli-FX™ EndoAnchor™ system to treat a wider range of patients with short, hostile aortic neck anatomies, independent of renal stenting.
 According to estimates from physicians across Europe and the United States, more than one-third of patients have AAA proximal neck anatomies less than 10mm[2]. Up to 30 to 40% of patients are considered unsuitable candidates for conventional EVAR[3] and more complex endovascular treatments offer a limited solution.
According to estimates from physicians across Europe and the United States, more than one-third of patients have AAA proximal neck anatomies less than 10mm[2]. Up to 30 to 40% of patients are considered unsuitable candidates for conventional EVAR[3] and more complex endovascular treatments offer a limited solution.
The FDA approval and CE Mark are supported by a short neck cohort of the ANCHOR registry, a global multi-center, multi-arm, prospective, post-market registry evaluating the real-world applicability of the Heli-FX™ EndoAnchor™ system. The ANCHOR registry is led by Co-Principal Investigators William Jordan, Jr., M.D., professor of surgery and chief, Division of Vascular Surgery and Endovascular Therapy at Emory University School of Medicine and Jean-Paul de Vries, M.D., Chief of Vascular Surgery at St. Antonius Hospital in Nieuwegein, the Netherlands. The short neck cohort one-year outcomes from a sub-analysis of 70 patients with proximal AAA neck lengths less than 10mm and down to 4mm, who were treated with Endurant™ II/IIs and Heli-FX™ system, demonstrated a technical success rate of 88.6%, based on delivery and deployment of the stent graft and each EndoAnchor implant, a 97.1% procedural success rate (investigator-assessed), with a rate of 1.9% Type Ia endoleaks at 1-year post-op (Corelab-assessed). At one-year there were no instances of AAA expansion or main body migration and through one year, no instances of AAA rupture. The overall secondary endovascular procedure rate was 4.7%. There was minimal EndoAnchor implant time added to the overall procedure, with an average of 17 minutes.
 Medtronic’s Heli-FX™ EndoAnchor™ systems feature an endovascularly-deployed anchor designed to attach a variety of aortic endografts to the native vessel wall. This off-the-shelf, customized solution minimizes the need for complicated procedures for the select subset of patients who would benefit from supplementary fixation including patients with challenging anatomies, risk factors for a secondary intervention, existing seal complications, as well as in situations where a physician may intraoperatively determine the need for additional security.
Medtronic’s Heli-FX™ EndoAnchor™ systems feature an endovascularly-deployed anchor designed to attach a variety of aortic endografts to the native vessel wall. This off-the-shelf, customized solution minimizes the need for complicated procedures for the select subset of patients who would benefit from supplementary fixation including patients with challenging anatomies, risk factors for a secondary intervention, existing seal complications, as well as in situations where a physician may intraoperatively determine the need for additional security.
This indication expands Endurant™ II/IIs stent graft’s hostile neck therapies. In December 2016, Medtronic also received CE mark for the Endurant™ II/IIs stent graft system to treat abdominal aortic aneurysm (AAA) patients using a ChEVAR procedure. This treatment is not approved in the US. The original Endurant™ stent graft system received the CE (Conformité Européenne) Mark in June 2008 and approval from the FDA in December 2010. The Heli-FX™ system is cleared by the FDA for distribution in the United States and has been granted CE Mark for distribution in the European Union. Both products are also commercialized in other countries worldwide.
Endovascular treatment requires lifelong, regular follow-up. Important Safety Information
UC201804838IE
References:
[1] September 2017
[2] Medtronic data on file.
[3] Perspect Vasc Surg Endovasc Ther. 2009;21:13-18.













