Biotronik today announced the limited release of its Oscar (One Solution: Cross. Adjust. Restore) multifunctional peripheral catheter and start of promotional activities at this year’s Leipzig Interventional Course (LINC 2023; 6–9 June, Leipzig, Germany).
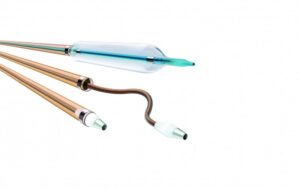
As indicated per Instructions for Use, the Oscar catheter is intended for dilation of stenotic segments in peripheral vessels. The device is comprised of three fully user-adjustable components (support catheter with integrated Lock Grip, extendable dilator and length-adjustable percutaneous transluminal angioplasty [PTA] balloon). It was developed to provide support during access into and to dilate stenoses in femoral, popliteal and infrapopliteal arteries.
The multifunctional system will be featured in the first symposium focused on the Oscar catheter at LINC 2023, with presentations from Koen Deloose (AZ Sint-Blasius, Dendermonde, Belgium), Peter A Schneider (University of California San Francisco, San Francisco, USA), Ralf Langhoff (Sankt Gertrauden-Krankenhaus, Berlin, Germany) and Mariano Palena (Maria Cecilia Hospital, Cotignola, Italy) on Wednesday 7 June at 12:30pm in Main Arena 1 during the session “Versatility without compromise: The peripheral arterial disease [PAD] workflow of the future with a new all-in-one solution”.
The first commercial procedure with an Oscar catheter in the USA was performed by Schneider. “With the Oscar catheter, I have a platform, support, pushability and crossability that I don’t have by cobbling together multiple other catheters and wires,” he commented.
Christoph A Binkert (Kantonsspital Winterthur, Winterthur, Switzerland) performed the first commercial procedure using an Oscar device in Europe. He remarked: “The Oscar catheter undoubtedly reduced several procedural steps and device exchanges. The ability to dilate the full lesion length followed by focal areas of residual stenosis with the same balloon was a great benefit to my procedural flow.”
The device began its initial commercial roll out in both the USA and Europe in late April.
“We are eager to get the Oscar catheter into the hands of peripheral interventionalists around the world and help to potentially change the way they approach difficult cases both above and below the knee,” said Joerg Pochert, President Vascular Intervention at Biotronik.
The Oscar multifunctional peripheral catheter will continue commercial expansion in the USA and CE mark-accepting regions over the coming months, a company press release advises.













